|
Project |
Micro-Systems & Control
Laboratory, NTHU |
Cell patterning Labchip for tissue engineering applications
Liver on a chip
![]() Objectives:
Objectives:
End-stage
organ failure or tissue loss is one of the most disastrous and costly issues in
medicine. Up to date, hundreds of millions of patients still suffer from
disease��s torment and over million surgical procedures are estimated to be
performed each year to treat these disorders worldwide. Over the last 50 years,
transplantation of a wide variety of tissues, reconstructive surgical
techniques, and replacement with mechanical devices have significantly improved
patient outcomes and become the most effective treatment. Unfortunately, organ
and tissue transplantation are imperfect solutions because surgical
reconstruction undergoes a critical problem due to lacking of abundant suitable
organs and tissues form limited and available human donors, which greatly
restrict the therapeutic results of surgery. In the year of 1987, J. Vacanti
and R. Langer coin a promising fashion named ��Tissue Engineering�� and aim at
ending up the problem owing to the limited source of donor��s organs. Tissue
engineering is an interdisciplinary field that applies the principles and
methods of engineering and the life sciences and targets at regenerating
natural tissues and regenerating artificial organs toward the development of
biological substitutes that could restore, maintain, or improve tissue and
organ function for human transplantation. In the tissue engineering, porous scaffolds
combined with adequate target cells and culture method have been used to
provide with physical and chemical cues for the cell culture, attachment,
proliferation, differentiation into functional tissues. Although advanced
biodegradable scaffolds, which morphologically mimic the human tissue, are
developed to mainstream biomaterial used as microenvironmental
matrix for the cell culture, it is still insufficient to guide, place and
distribute the heterogeneous cells to reconstruct complicated architectures of complex
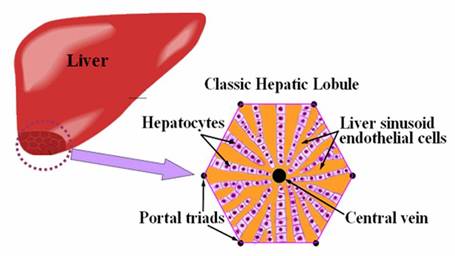
tissue especially like kidney and liver. In particular, hepatic sinusoids, the
special liver��s micro-vascular systems, which are lined by liver sinusoid
endothelial cells to form a radiate pattern, are essential for normal liver
functions and hepatocyte survival (Fig 1.). Thus,
how to develop an effective cell manipulating method that could achieve adequate
positioning of both hepatic
and endothelial cells to reconstruct the complex liver tissue according to its native
architecture has become the important and challenging issue in liver tissue
engineering.
|
|
|
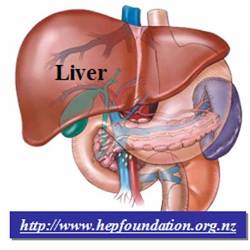
Fig.
1. Illustration of liver organ. The enlarged and cross-sectional view shows
one unit of the liver tissue, the classic hepatic lobule. |
![]() Technical
Approach:
Technical
Approach:
To reconstruct the appropriate heterogeneous pattern of the classic hepatic lobule consisting of hepatic and endothelial cells and to reserve intimate cell-cell contact to modulate interactions for further cell behaviour are the challenged and important steps toward the development of successful artificial liver and highly relies on the cell patterning technique. Fine cell patterning techniques capable of precisely controlling cell position provide the basis ability for rebuilding cell blocks; play a crucial role in the field of tissue engineering, as well as cell-based biosensors, medical diagnostics, and drug delivery. In recent years, a variety of advanced progresses in tissue engineering have been dedicated to the developments of manifold cell patterning techniques for tissue engineering applications, such as photolithography, microcontact printing, microfluidic patterning, laser-guide direct writing, ink-jet printing and dielectrophoresis (DEP). Among these cell-patterning approaches, electric-field induced DEP effect offers the capability of active and in-parallel cell-manipulation to rapidly achieve precise positioning of heterogeneous cell populations with high cell viability and high patterning resolution, which is a superior candidate considering to be applied to the field of Tissue engineering. For this goal, a rapid heterogeneous-cells patterning microfluidic chip via the enhanced DEP trap is designed and demonstrated to facilitate the reconstruction of lobule-mimetic liver tissue in vitro (Fig. 2). By taking advantage of the novel design of concentric-stellate-tips-electrode, which simulated and analyzed via CFDRC, the enhanced inhomogeneous field-field could be generates in spatial to precisely snare plenty of individual cells to form the desired lobule-mimetic pattern via the high-precision DEP manipulation (Fig. 3). With the novel design and materialization of this materialized microfluidic chip design (Fig. 4), the original randomly distributed liver cells in our chip could be manipulated in parallel and align into desired radiate pearl-chain array to form the lobule-mimetic radiate pattern, mimicking from the morphology of real liver tissue, with good cell viability after cell-patterning DEP manipulation. Heterogeneous integration of liver-cell patterning is demonstrated on our microfluidic chip with several thousands of HepG2 cells and HUVEC cells snared and patterned on the patterning area of about one millimeter scale (Fig. 5). High-resolution cell pattering methods capable of controlling the heterogeneous cells as well as the cell-cell interactions, which potentially modulate the cell behavior, would enable reproducible control over the cellular microenvironment and could benefit the maintenance of cell functions in vitro physiological systems. Based on our knowledge, this research reports the first result of lobule-mimetic liver tissue reconstruction in-vitro. This proposed cell-patterning chip demonstrates the rapid in-parallel heterogeneous patterning of live liver cells via a novel enhanced DEP trap design inside the microfluidic chip and potentially could be applied for the further studies in biological research, biomedical investigation and tissue engineering. For more detail, please see our paper published at the journal of Lab on a Chip, 2006, 6(6), pp. 724-734. Furthermore, this paper is honorably selected as the ��Hot article�� and the ��inside cover�� at the issue of 2006, 6 of Lab on a Chip. Also, a research highlight is received and announced by RSC Chemical Biology at June, 7, 2006 as shown in the Fig. 6 below.
|
|
|
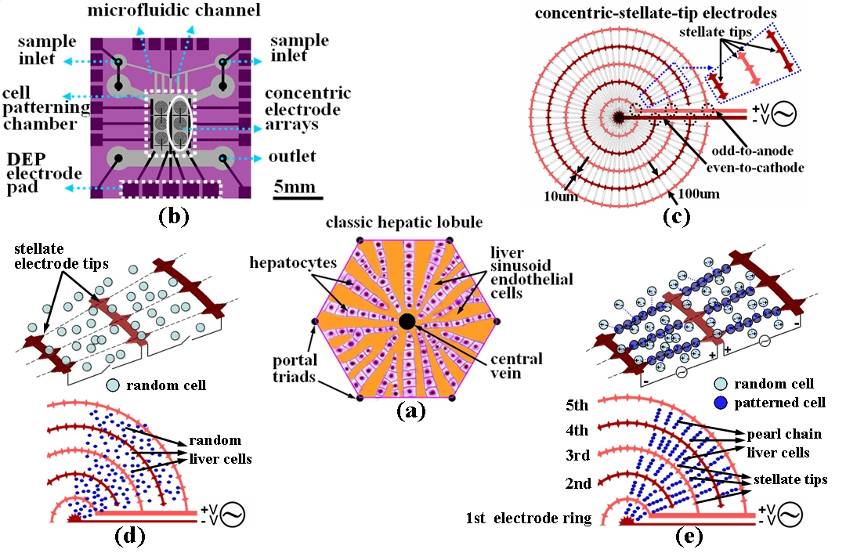
Fig. 2. The illustration,
chip design and principles for the liver-cell patterning microfluidic chip.
(a) The illustrated
configuration for one unit of the classic hepatic lobule. (b) Top schematic view of the cell-patterning microfluidic
chip. This chip is composed of a transparent glass substrate embedded with
the matrices of concentric-stellate-tip array electrodes and a PDMS top
cover. (c) The enlarged view for a unit of concentric-stellate-tip array
electrodes. (d) Liver cells are spatially randomly distributed before the ac DEP voltage is applied. (e) The
energized concentric-stellate-tip array electrodes provide well-defined local
electric-field maxima. Liver cells are snared and
aligned along the field-induced orientation to form the radiate
pearl-chain two-dimensional patterns due to the positive DEP after the ac DEP
voltage is applied. |
|
|
|
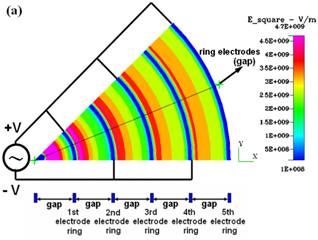
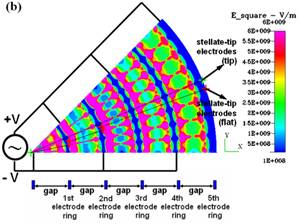
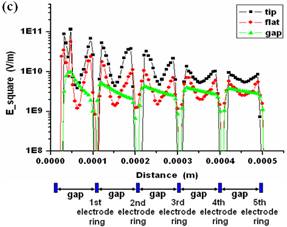
Fig.
3. CFDRC numerical simulation of DEP for the concentric-ring-array electrodes
and for the concentric-stellate-tip array electrodes. (a) The distribution of
the root mean square of ac electric field (E_square) for the
concentric-ring-array electrodes. (b) The distribution of E_square for the
concentric-stellate-tip-array electrodes. (c) The numerical comparison for
the E_square versus the radial distance from the center of ring circles. |
|
|
|
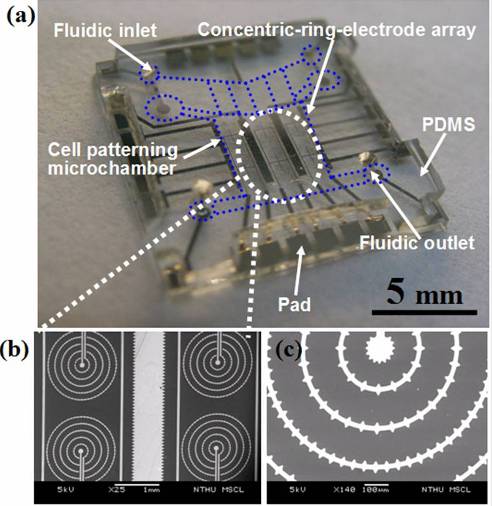
Fig. 4. Materialization of cell-patterning
microfluidic chip. (a) The final cell-patterning chip. (b) The SEM image shows
the concentric-stellate-tips-electrode array. (c) The close-view of (b) shows
the detail geometry of concentric ring electrodes with regular stellate-tips. |
|
|
|
Fig. 5.
On-chip heterogeneous-cells patterning demonstration.
(a) The first
group of HepG2 cells
are snared and patterned in radiate pearl-chains via the concentric-stellate-tip
array electrodes with the in-parallel DEP manipulation.
(b)
The on-chip demonstration for the rapid heterogeneous-integration patterning
of HepG2 cells
(green
fluorescence)
and HUVECs (red fluorescence). Both
HepG2 pearl-chains and HUVECs
pearl-chains are snared and align in the radiate patterns via the enhanced field-induced DEP
manipulation. The alternate radiate patterned HUVECs mimic
the shape and the function of sinusoid-like vascular endothelial lining cells
that are shown in the classic real hepatic lobule. (c) The fluorescent
control group
with the two
types of cells randomly distributed over the
cell-patterning chamber without
DEP manipulation. |
|
|
|
Fig. 6. The ��inside cover�� of the journal of Lab on a chip at the issue of 2006, 6 and a research
highlight is received and announced by RSC Chemical Biology at June, 7, 2006 |
![]() References:
References:
1. C. T. Ho, R. Z. Lin, W. Y. Chang, H. Y. Chang and C. H. Liu, "Rapid heterogeneous liver-cell on-chip patterning via the enhanced field-induced dielectrophoresis trap" , Lab Chip, 6, pp.724 - 734, (2006) (selected as Inside Cover and Hot Article of Lab on a chip Journal (2007 SCI Impact Factor: 5.8) and highlighted by RSC Chemical Biology, June 7, 2006.)
2. Ruei-Zeng Lin, Chen-Ta Ho, Cheng-Hsien Liu, and Hwan-You Chang, ��Dielectrophoresis-based cell patterning for tissue engineering,�� Biotechnology Journal, 2006, 1, 949-957, (one of the three papers highlighted at "In the Issue" section)
3. Chen-Ta Ho and, Po-Chi Lin, Hwan-You Chang, and Cheng-Hsien Liu, ��A Cell-Patterning Biochip Based on Dielectrophoresis for liver tissue application,�� Proceedings of 9th International Conference on Miniaturized Systems for Chemistry and Life Sciences, October 9, 2005, Boston, USA, pp. 1368-1370, 2005. (MicroTAS 2005)
Collaboration:
Contact Information:
�P
Chen-Ta Ho d917725@oz.nthu.edu.tw
�P
Chien-Yu Chen g9533541@oz.nthu.edu.tw
�P
Song-En
Gong g9535501@oz.nthu.edu.tw
�P
Cheng-Hsien Liu liuch@pme.nthu.edu.tw